- Brother controlcenter3 conflicting with controlcenter4
- How to jailbreak xbox 360 slim
- Pokemon platinum rom file
- Desitv serial
- Sql maestro php generator for mysql wizard
- Danganronpa junko
- Parametric sensitivity frontline solver example
- Free electra x vst
- Ipad pro amaziograph
- Nikon capture nx2 tutorial youtube
- Lemonade tycoon best recipe
- Dorks para sqli dumper
- Whisperer in darkness
- Pc error 1935 microsoft vc80 atl type win32 windows 10
- Install vr xbox 360 bios
- Common ion effect on solubility pogil key
- What is 3d viewer and why do i need it
- Yamaha audiogram 3 free driver software
- Progressions for french cafe music
- Alien obduction
- Darkfall unholy wars crafting spreadsheet
- Microsoft access product returns database template

Any further addition of salt will remain undissolved: Saturated solutionĭ) The product of the molarities of the dissolved ions, raised to a power equal to the ion's coefficient in the balanced chemical equation : Solubility product constantĮ) The maximum number of moles of a salt that will dissolve in 1 L of solution : Molar solubility This system described here will contain HNO2, water and NaNO2Ī) A decrease in the solubility of an ionic compound as a result of the addition of a common ion : Common ion effectī) The mass of a salt in grams that will dissolve in 100 mL of water: SolubilityĬ) A solution that has dissolved the maximum amount of a compound at a given temperature.

Also, the extent of ionization of HNO2 in a system that already contains NaNO2 will be decreased compared to its extent ionization in pure water. If I try to dissolve NaNO2 in a solution of HNO2, the solubility of NaNO2 in the HNO2 solution will be less than its solubility in pure water due to common ion effect. We refer to the phenomenon described above as common ion effect. If I dissolve AB in water and form a solution, subsequently, I add solid CB to this solution, the solubility of CB in this solution is found to be lees than the solubility of CB in pure water because of the ion B^- which is common to both substances in solution. Both CB and AB are soluble in water to a certain degree as shown by their respective KSp. Secondly, I have another ionic distance with cation C and anion B, its chemical formula is CB.
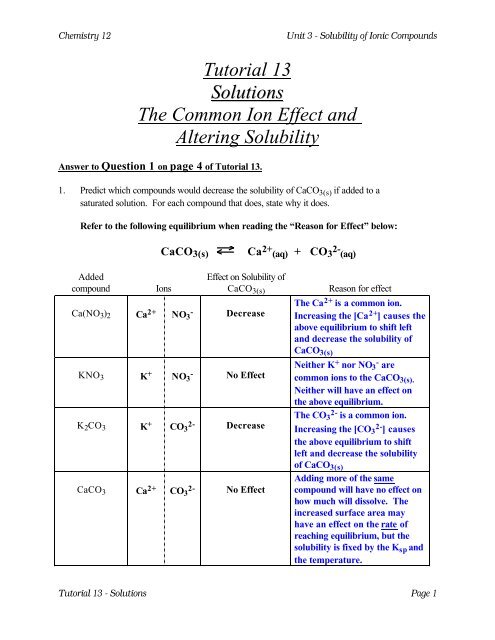
Assuming I have a solution of an ionic substance that contains a cation A and an anion B, this ionic substance has chemical formula AB. In order to understand exactly what common ion effect is, let us consider a simple unambiguous example. Common ion effect refers to the decrease in the solubility of a substance in a solution with which it shares a common ion.
